Security and compliance
Ensure data security and compliance with data masking, monitoring, and change traceability
Redgate Test Data Manager scans your databases, identifies PII automatically, and masks it with realistic alternatives across SQL Server, Oracle, PostgreSQL, and MySQL. From discovery to compliant test data in minutes, not months.
“Data Masker provides truly representative copies of the production database which are a fraction of the size of the original, with the sensitive data masked. It also integrates with SQL Data Catalog, which can provide the masking set necessary to protect sensitive data.”







Masking sensitive data across your estate enables you to deliver secure data to downstream environments in non-production databases for development and testing.
Enabling this automatically ensures your data is secure across your organisation – therefore minimising data risk, and ultimately protecting your business.
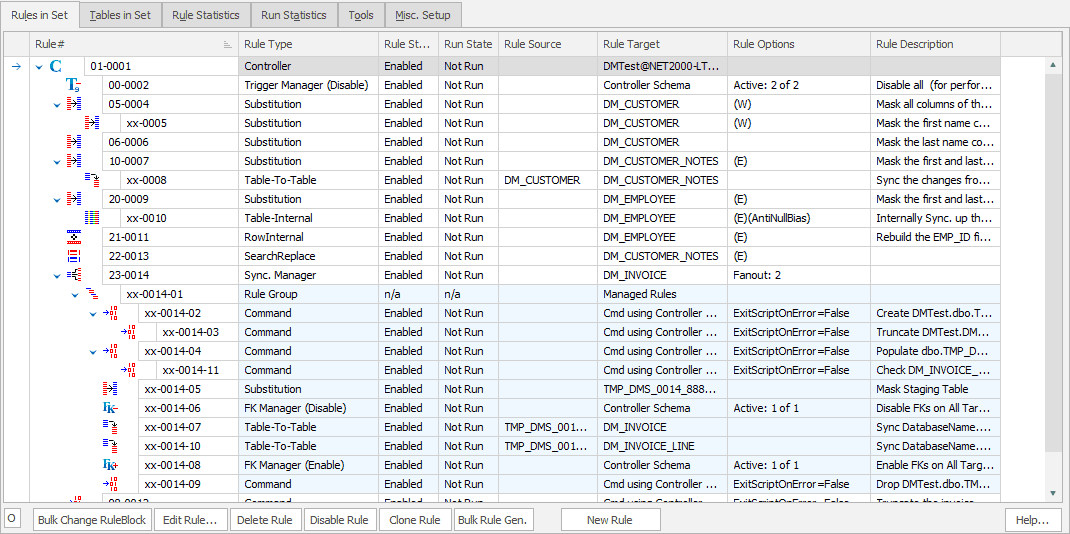
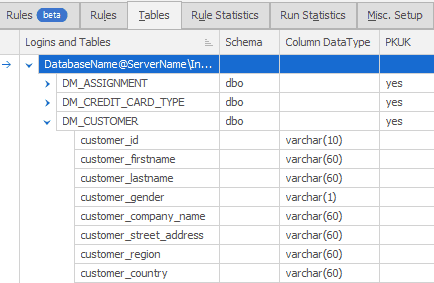
Remove sensitive data from your test and development systems
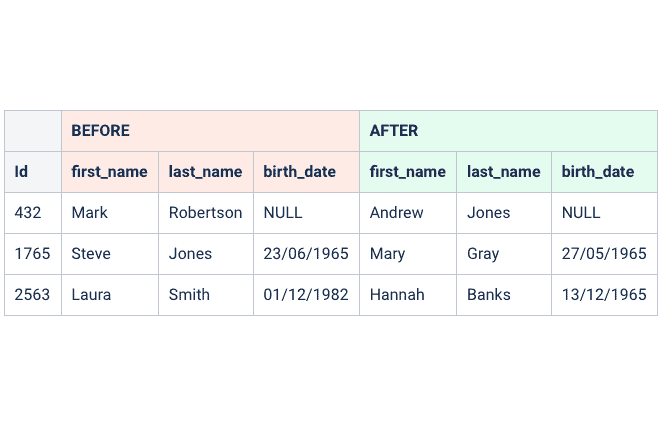
System provides realistic data when masking
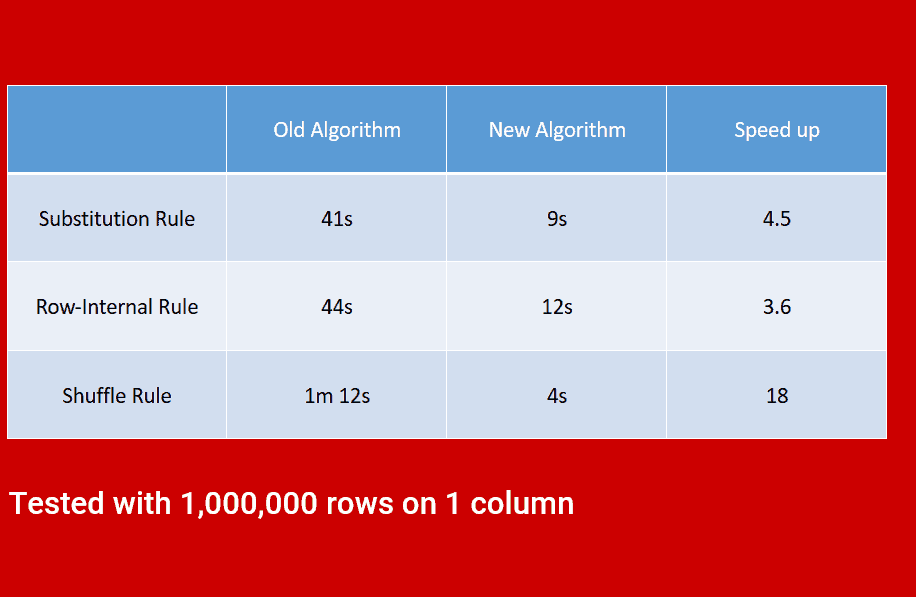
High performance system, enabling a quicker time to masking

System enables automated masking for future compliance
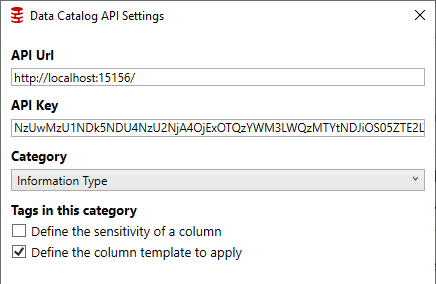
Ability to integrate with existing processes with both Data Catalog and Clone products
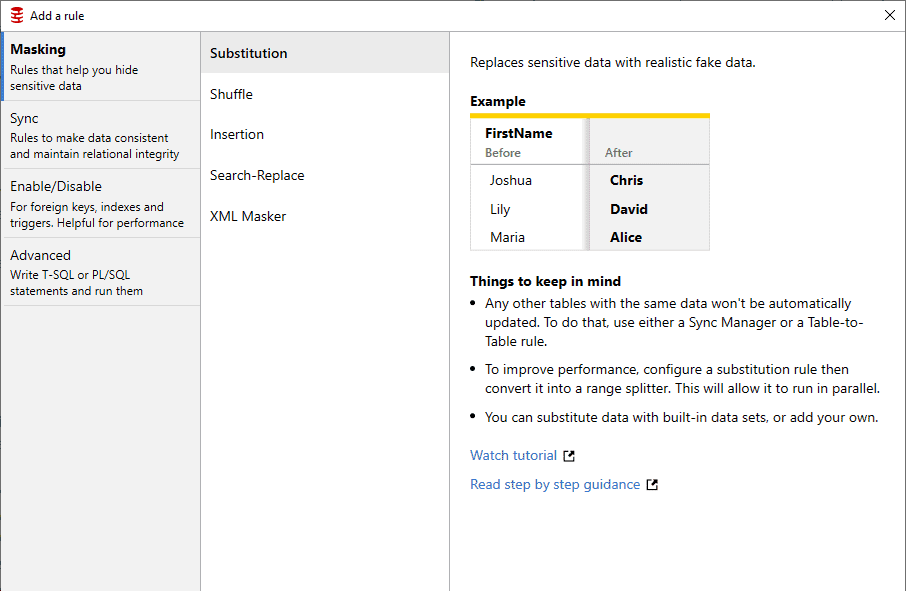
Ease of use for non-specialist users

University
Learn the basics of Data Masker with self-paced online training courses from Redgate's Microsoft Data Platform MVPs and engineers.
Blog
Republic Bank strive to stay ahead of their competitors when it comes to technology, but with over 1900 databases full of sensitive information, they have serious data masking and cataloging requirements.
Get started with Data Masker
Start masking sensitive data today – build your data masking plan with help from our Support team, and get started with the comprehensive tutorials, sample tables and masking rules included in the fully functional trial.

If you'd like any help, or have a question about our tools or purchasing options, just get in touch.